This method, however, faces numerous challenges among them being lack of reference standards which compromises on its reproducibility and effectiveness. In a previously published literature, the Ni 2+/Ni 3+ ratio has been approximated using the Gaussian/ Lorentzian curves. 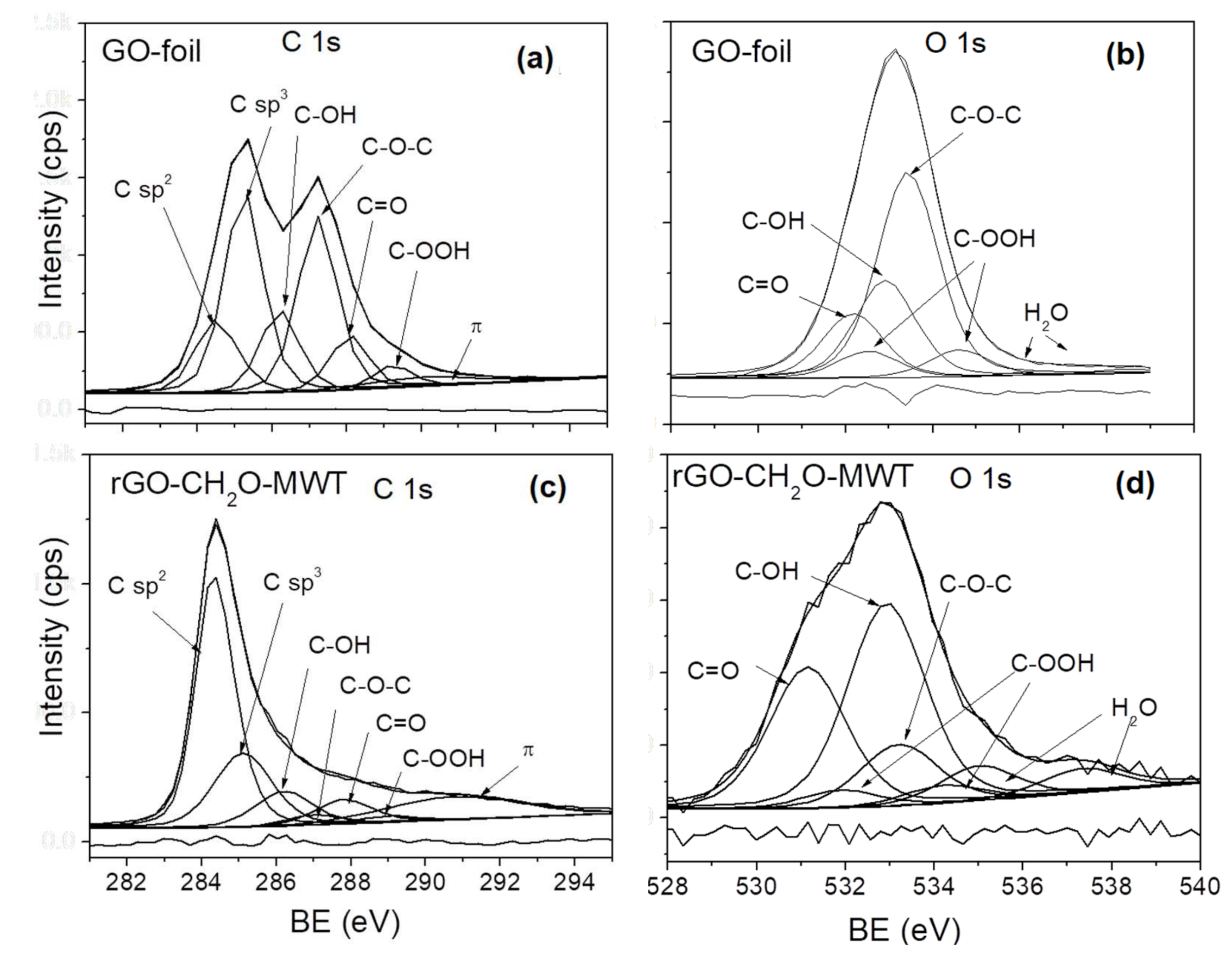
However, the Ni 2+/Ni 3+ ratio has not been fully explored. Owing to high uncertainty degree in chemical states quantification, numerous fitting methods and interpretations of X-ray photoelectron spectroscopy spectra have been proposed. It has been of great importance in the evaluation of the crystal structure and electrochemical performance of the lithium-nickel-cobalt-manganese oxides. 
X-ray photoelectron spectroscopy(XPS) technique has been widely used in the compositional and chemical state analysis. To that note, researchers have been looking for efficient ways of controlling the Ni 2+/Ni 3+ ratio during the synthesis and storage phases. During synthesis and storage, the presence of unstable cation in the solid state may result in the reduction of trivalent ions to divalent ions thus leading to an increase in the cation ratio which may, in turn, reduce their electrochemical performance. Unfortunately, nickel ions in these oxides exist as divalent or trivalent states. They exhibit high energy density, discharge capacity and relatively low cost. Recently, the use of Ni-rich lithium-nickel-cobalt-manganese oxides as cathodes in lithium-ion batteries has attracted significant attention amongst researchers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
